Structural basis for the inhibition of cGAS by nucleosomes
- Sep 11, 2020
- 2 min read
Updated: Sep 18, 2020
Tomoya Kujirai, Christian Zierhut, Yoshimasa Takizawa, Ryan Kim, Lumi Negishi, Nobuki Uruma, Seiya Hirai, Hironori Funabiki, Hitoshi Kurumizaka
Published in Science 10, Sep 2020 (Link)
Introduction
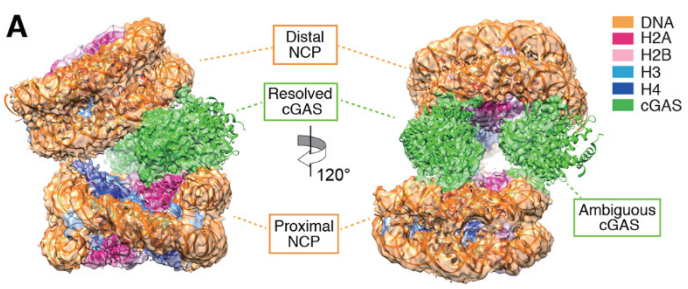
cGAS is a major innate immune sensor of pathogen DNA. While pathogen DNA can activate cGAS signaling, under normal conditions, self-DNA does not. We previously hypothesized that the organization of self-DNA into chromatin makes it refractory to cGAS and underlies self vs non-self discrimination by cGAS. In this study, we describe the structural basis for how cGAS is held inactive when bound to nucleosomes, the fundamental building block of chromatin.
Highlights
- They cryo-EM structure of the human cGAS:nucleosome core particle (NCP) complex explains how cGAS activation by chromosomal self-DNA is prevented
- In the complex, two cGAS promoters are sandwiched between two nucleosome core particles (NCPs)
- Within the complex, cGAS dimerization is prevented, and all three DNA binding sites on cGAS are blocked
- Biochemistry confirms that nucleosome-binding underlies the inhibition of cGAS by nucleosomes
Summary
The cyclic GMP-AMP synthase (cGAS) senses invasion of pathogenic DNA and stimulates inflammatory signaling, autophagy and apoptosis. Organization of host DNA into nucleosomes was proposed to limit cGAS autoinduction, but the underlying mechanism was unknown. Here, we report the structural basis for this inhibition. In the cryo-EM structure of the human cGAS-nucleosome core particle (NCP) complex, two cGAS monomers bridge two NCPs by binding the acidic patch of H2A-H2B and nucleosomal DNA. In this configuration, all three known cGAS DNA-binding sites, required for cGAS activation, are repurposed or become inaccessible, and cGAS dimerization, another prerequisite for activation, is inhibited. Mutating key residues linking cGAS and the acidic patch alleviates nucleosomal inhibition. This study establishes a structural framework for why cGAS is silenced on chromatinized self-DNA.





Comments